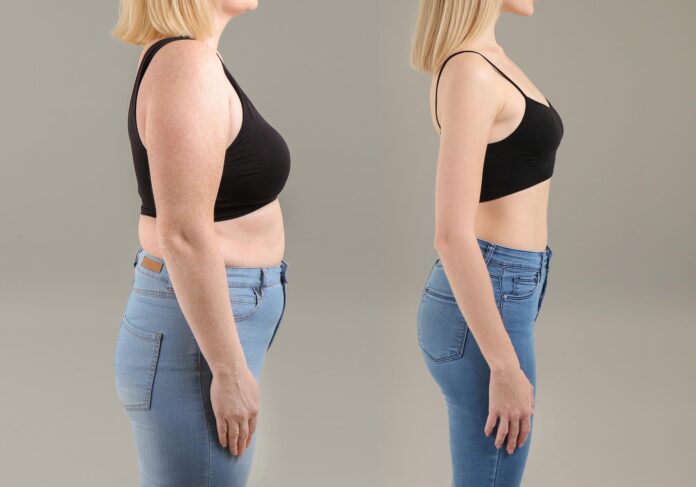
A promising new drug candidate has shown the ability to prevent obesity and severe liver damage in mice, even when the animals consume a diet high in sugar and fat. The treatment works by targeting a specific mechanism inside mitochondria—the energy-producing powerhouses of cells—specifically by regulating how magnesium moves within these structures.
Researchers from The University of Texas Health Science Center at San Antonio (UT Health San Antonio), along with collaborators from the University of Pennsylvania and Cornell University, published their findings in Cell Reports. The study highlights an unexpected link between magnesium transport and metabolic health, suggesting that controlling this flow could be a key to treating obesity, fatty liver disease, and type 2 diabetes.
The Role of Magnesium in Metabolism
Magnesium is a vital mineral, essential for regulating blood sugar, blood pressure, and bone health. It is the fourth most abundant cation in the human body, following calcium, potassium, and sodium. However, the study reveals that its location within the cell matters significantly.
Inside mitochondria, magnesium appears to act as a regulator of energy production. The research suggests that when too much magnesium enters mitochondria through a channel controlled by the MRS2 gene, it can slow down the cell’s ability to efficiently burn sugar and fat.
“It puts the brake on, it just slows down,” explained Travis R. Madaris, a doctoral student and co-lead author of the study.
To test this theory, researchers removed the MRS2 gene in a group of mice and fed them a “Western diet”—rich in sugars and fats—for up to 52 weeks. While control mice gained significant weight and developed metabolic issues, the mice without the MRS2 gene remained lean. Despite eating similar amounts of food and engaging in comparable physical activity, these mice exhibited a more active metabolism. Their white fat tissue began to “brown,” shifting toward a more energy-burning type of fat, and they efficiently utilized sugar and fat for energy.
Protection Against Liver Disease
The benefits extended beyond body weight. The liver results were particularly striking. In typical mice, the high-fat, high-sugar diet led to fatty liver changes, fibrosis, enlargement, and frequent tumor formation. In contrast, mice lacking the MRS2 gene showed no evidence of fatty liver disease and maintained healthier blood glucose levels.
The researchers discovered that reducing magnesium entry into mitochondria altered how cells handled citrate, a molecule used to produce fat. With less mitochondrial magnesium, less citrate left the mitochondria to be converted into fat. This shift likely explains why these mice accumulated less fat in both their liver and body, avoiding the metabolic syndrome markers typically associated with poor diet.
A Drug Mimics Genetic Changes
Building on these genetic findings, the team developed a small-molecule compound called CPACC, which blocks magnesium transport through the same mitochondrial pathway. UT Health San Antonio has filed a patent application for this drug.
In experiments, CPACC demonstrated several beneficial effects:
– Reduced lipid buildup in liver cells.
– Increased mitochondrial respiration.
– Lowered plasma citrate levels.
– Promoted the development of “beige fat,” a metabolically active form of fat.
When mice on a high-fat diet were injected with CPACC every three days for six weeks, they experienced limited weight gain and improved liver function markers.
“Lowering the mitochondrial magnesium mitigated the adverse effects of prolonged dietary stress,” said Manigandan Venkatesan, PhD, a postdoctoral fellow and co-lead author.
Future Implications and Next Steps
These findings suggest that mitochondrial magnesium channels could be a powerful new target for treating cardiometabolic conditions, including obesity, type 2 diabetes, and fatty liver disease. However, researchers emphasize that this is early-stage work.
The current studies were conducted on mice using a global knockout of the MRS2 gene, meaning the channel was removed in all tissues. Future research will need to develop safer, more refined modulators that can target specific tissues without disrupting magnesium’s essential roles elsewhere in the body.
“A drug that can reduce the risk of cardiometabolic diseases such as heart attack and stroke, and also reduce the incidence of liver cancer, which can follow fatty liver disease, will make a huge impact,” said senior author Madesh Muniswamy, PhD, director of the Center for Mitochondrial Medicine at UT Health San Antonio.
Broader Scientific Context
Recent studies further support the importance of mitochondrial magnesium transport beyond obesity. A 2024 study in Mitochondrion showed that lower magnesium levels in mitochondria increase the activity of the mitochondrial calcium uniporter, influencing calcium signaling and cell stress. Additionally, a 2026 study in Hypertension linked the MRS2 channel to pulmonary arterial hypertension, finding that reducing its activity improved mitochondrial function in diseased cells.
While human trials are still ahead, this research opens a new avenue for understanding how cellular energy regulation can be manipulated to combat diet-related diseases.